Cleaning and Corrosion Protection with Solvents
Protect parts against rust in an efficient and sustainable way.
It is the task of industrial parts cleaning, both during and after manufacturing, to fulfill surface cleanliness specifications that are required for further processing or workpiece assembly. Depending on the contamination, non-halogenated hydrocarbons and modified alcohols demonstrate definite advantages. They not only ensure reliable, efficient and sustainable removal of oils, coolants and chips, but also allow for performing cleaning and corrosion protection processes—even long-term protection—in one machine.
Metallic parts inevitably get dirty during manufacture and machining operations. Foreign matters such as coolant residue, as well as chips and particles, normally become a problem in downstream processing such as heat treatment, welding, coating or assembly. In addition, contaminants may impair the quality, functionality and service life of the finished product.
Featured Content
Thus, industrial parts cleaning is an indispensable step in the production chain. The most frequently used technique is wet-chemical cleaning.
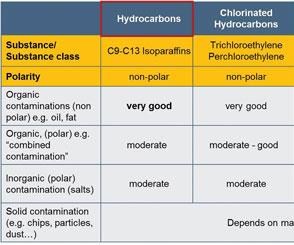
‘Like Dissolves Like’ Principle
Process effectiveness as well as quality, cost efficiency and stability of the cleaning process are critically dependent on the dissolving capacity of the cleaning agent employed. Both aqueous detergents and solvents are commonly used. In selecting the right cleaning medium, the “like dissolves like” principle of chemistry, based on the polarity of a substance, applies. It indicates that a contaminant dissolves best in a solvent (cleaning agent) that has a similar structure to itself (see Table 1). In other words, water-based (polar) types of contamination such as coolant and lubricant emulsions, polishing pastes, additives and salts are typically cleaned with aqueous cleaning agents (hydrophilic).
For removing non-polar contaminants such as lubricating and cooling oils, greases and waxes, a solvent (lipophilic) is usually the preferred cleaning agent. Solvents are commonly classified into chlorinated hydrocarbons (CHC), non-halogenated hydrocarbons (NH) and modified alcohols, also known as polar solvents (P).
As modified alcohols offer lipophilic and hydrophilic properties, they are effective in removing non-polar (lipophilic) and polar (hydrophilic) contaminations. Thus, they are used as an alternative to hydrocarbons as well as to aqueous detergents.
Hydrocarbons and Modified Alcohols
Health, safety and environmental concerns have negatively affected the reputation of industrial parts cleaning with solvents. However, if a cleaning task can be performed with an aqueous detergent or a solvent alike, using non-halogenated hydrocarbons or modified alcohols (hereinafter also referred to as solvents or hydrocarbons) offers substantial advantages:
Good degreasing quality.
Solvent recovery via distillation. Both hydrocarbons and modified alcohols are distillable, which means that oils and emulsion brought into the machine and mixed with the solvent are removed continuously and automatically, even if the oil input is high. This process ensures constant good solvent quality and hence, consistent cleaning quality. Additionally, the continuous reconditioning allows for a long solvent life span and low consumption.
No quality control necessary. In contrast to aqueous cleaning, where pH level, detergent concentration, conductivity, and so on, have to be checked regularly, solvent cleaning does not require continuous quality controls. An exception is when chlorinated oils, used in such processes as stamping and deep drawing, are brought into the machine. In such cases, solvent distributors offer adequate test equipment and stabilizers that might be required.
Compatible with almost all materials. Unlike aqueous detergents, solvents do not cause oxidation, discoloration, burning, dulling or other surface impairments. As a result, chemical surface effects such as etching are not possible, however, these are not actual cleaning jobs.
100 percent dry parts. Solvents allow for drying parts completely, even in blind holes and hidden passageways, without the use of hot air.
Low energy consumption and running costs. Both heating up the cleaning fluid and drying the parts require less energy in a solvent cleaning process. In addition, the bath does not need to be controlled and changed regularly. The cost of consumables such as cleaning agents, and fresh and waste water, as well as waste disposal costs, are usually lower, too.
Short- and long-term corrosion protection. Modern solvent cleaning machines allow for performing cleaning and corrosion protection steps in one system.
Vacuum Technology
The concerns already mentioned about solvents in industrial parts cleaning are certainly justified with regard to open-top vapor degreasing systems, which sometimes are still in use. Even so-called “closed” systems that are equipped with cover plates and cooling coils as a condensation trap for emission reduction are not considered ideal solutions regarding HSE. Additionally, the solvent consumption is much higher when compared with modern, fully closed and safe vacuum cleaning machines.
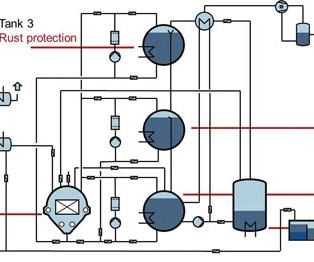
These systems are equipped with a working chamber and automatic feeding system, one or more tanks for cleaning and corrosion protection, and an integrated vacuum distillation system. They operate under full vacuum—cleaning, degreasing, drying, and, if applicable, corrosion protection are performed, in reduced pressure atmosphere in one system. This design results in several benefits.
No Hot Air or Waste
One benefit of these systems is the ability to dry parts 100 percent without energy-intensive, generated hot air. The use of a vacuum enables the hydrocarbon to dissolve from the liquid into the gaseous phase completely at low temperature. Furthermore, it reduces the saturated vapor pressure, which enhances the capillary transport of the solvent. As a result, even the smallest blind holes and passages dry easily.
Low consumption of hydrocarbon is achieved through continuous vacuum distillation and solvent treatment. Separated oil is automatically discharged from the system and collected in a waste drum for disposal after certain intervals. The regenerated solvent is led back into the tank for consecutive cleaning cycles. This design allows for the hydrocarbon to be used in a closed loop without complex and time-consuming solvent tests and any disposal of used cleaning media. Fresh water, disposal of waste water and separation of chemical substances are not required.
Operator and Environmental Safety
Fully closed vacuum parts cleaning machines eliminate the need for additional explosion protection when working with hydrocarbons. Because of the vacuum, the concentration of the gaseous mixture of air and solvent in the machine is shifted into a non-flammable zone. Furthermore, integrated cooling systems are used to extract solvent from exhaust air.
Also, machine operators are not in contact with the solvent. The parts to be cleaned are fed automatically into the working chamber. The solvent is not brought to the chamber until it is closed and sealed. When hydrocarbon needs to be added, this process also takes place automatically without operator interaction.
Passivation versus Rust Protection
Nowadays, it is not uncommon that parts such as those made of steel and cast iron travel thousands of miles by road, air or sea before they reach their point of use. Therefore, they must be effectively protected from corrosion. This is also true if corrodible parts get stored until their use. A fairly common method is to put the parts in a box, sometimes lined with a foil, and pour oil over them. Although it’s easy and fast, the oil only reaches external surface areas, while internal areas, especially of parts with complex geometries, are not protected and start to corrode.
Here are some alternatives.
Passivation of parts:
- Temporary or “short-term” corrosion protection is often carried out in water-based cleaning systems with an aqueous cleaning agent (neutral) that contains amines. With this method, a protection time of several hours to days can be achieved.
- With a separate passivation bath, the protection time can be prolonged to several weeks, depending on amine concentration.
- Using emulsified oils in water-based systems can stretch the protection time to months. However, this method includes the risk that the emulsified oils cannot be dried on the part surface, leading to cross-contamination. In case these oils are dried in a hot air dryer, they can be carried over, leading to a fire hazard. Moreover, as the protective film is created out of a two-phase system (water and oil), the layer on the part surface is often not homogenous and spots can occur.
Rust protection of parts:
- For “long-term” corrosion protection (protection time from weeks and months to years, depending on the media concentration) rust preventive media such as oils, greases and waxes are employed. As these media are 100-percent soluble in hydrocarbons and modified alcohols, they can be used in fully closed vacuum cleaning systems.
Solvent systems set up to perform rust protection are often equipped with a separate tank (such as a second or third food tank) that is used exclusively for this purpose. The rust protection tank contains hydrocarbon or modified alcohol plus the required amount of rust protection media. After the parts are cleaned, the work chamber is flooded from the rust protection tank with media, and then the corrosion protection process takes place. Like the cleaning process, it guarantees that all areas of the part are reached by the media. This process is followed by the vacuum drying process in which the solvent is transported back to the rust protection tank while the rust protection film remains on the part surface.
The protective film is generated from a fully soluble, salt-free, one-phase system, and the layer on the part surface is homogenous, completely closed and dry. It is not broken if the surface is touched or if it comes into contact with packaging material.
The choice of rust protection media depends on the machine technology and solvent used. The film thickness—usually between 2 and 5 microns—is influenced by the concentration of rust protection media in the solvent and the dripping off time. The dosing of the rust protection media can be done manually or automatically.
Protection Time, Concentration
Information on protection time and required concentration is commonly provided by the supplier of the rust protection media. Depending on the corrosion protection product, typical concentrations are between 0.5 and 25 percent. They are based on empiric tests in the laboratory for determining the film weight and density (percent in solvent) after detecting a specific calibration curve.
For determining film weight, rust protection oil is applied on a standard sheet metal (such as Q-Panel R35) in the recommended concentration. The sheet metal is weighed before and after this process.
Film weight is one measure to determine the approximate consumption of rust protection media with the specific part geometry. The other one is film thickness.
Film weight [g/m²] = mass (rust protection oil) [g] / area (test sheet metal) [m²]
Film thickness [µm] = film weight [g/m²] × 100 / density (rust protection oil) [g/cm³]
Film thickness is becoming an increasingly important scale unit because more and more customers of part manufacturers not only require the media as corrosion protection, but also as assembly oil. Therefore, the protection layer must be applied in a defined thickness.
Since the concentration should also be checked at the part manufacturer, various methods have been developed. Measurement via evaporation residue with a specific moisture analyzer is the most commonly used method. It requires a sample of approximately 2 gr (0.07 oz) of the solvent corrosion protection oil mixture. Inside the analyzer, the solvent gets evaporated while the remaining oil is measured as a percentage of weight. The concentration can then be determined with a conversion chart.
Another measure method for determining concentration is the refractive index. One drop of media is given into a manual, handheld refractometer. The refractive index is then viewed by the user through a magnifying eyepiece. As the refractive index is temperature dependent, it is important to use a refractometer with an integrated thermometer. Measurements can be performed with media at 68°F, 77°F and 86°F. Automated and highly sensitive operating measurement instruments are also used for density measurement.
Modern, fully closed cleaning systems are an efficient and safe way of performing cleaning and even long-term rust protection of parts in a single system. For global Tier 1-3 precision part manufacturers, this technology is well established.
RELATED CONTENT
-
Simple, Effective Parts Cleaning
After trying an array of parts cleaning methods over the years, this shop has implemented an environmentally friendly, relatively simple system to clean every part it produces.
-
A Primer On Parts Washing—Here’s How To Get Your Parts Really Clean
Cleaning is loosely defined as the process of removing unwanted contaminants or dirt from a surface. It does not alter the surface physically or chemically. A properly cleaned surface is just the same as it was prior to cleaning, except it is missing the dirt.
-
Cleaning Parts Cost Effectively
A shop should look at all aspects of the production process to maximize productivity. This includes selecting an efficient cleaning system.